Nickel transition metal Chemistry nickel(II) Ni2+ complex ions ligand substitution redox chemical reactions principal oxidation states +2 +3 GCE AS A2 IB A level inorganic chemistry revision notes

Nickel transition metal Chemistry nickel(II) Ni2+ complex ions ligand substitution redox chemical reactions principal oxidation states +2 +3 GCE AS A2 IB A level inorganic chemistry revision notes

solid state | L10 | Nickel oxide exist as Ni0.98 O 1.00. What fraction of Ni exist as Ni2+ and Ni3+ - YouTube

Ni(II)/Ni(III) redox couple endows Ni foam-supported Ni2P with excellent capability for direct ammonia oxidation - ScienceDirect

Cool trick Electronic Configuration of Ni , Ni+2 and Ni+3 || electronic configuration of nickel - YouTube
![MeCN)Ni(CF3)3]− and [Ni(CF3)4]2–: Foundations toward the Development of Trifluoromethylations at Unsupported Nickel | Inorganic Chemistry MeCN)Ni(CF3)3]− and [Ni(CF3)4]2–: Foundations toward the Development of Trifluoromethylations at Unsupported Nickel | Inorganic Chemistry](https://pubs.acs.org/cms/10.1021/acs.inorgchem.0c01020/asset/images/acs.inorgchem.0c01020.social.jpeg_v03)
MeCN)Ni(CF3)3]− and [Ni(CF3)4]2–: Foundations toward the Development of Trifluoromethylations at Unsupported Nickel | Inorganic Chemistry

Synthesis and Characterization of Three-Coordinate Ni(III)-Imide Complexes | Journal of the American Chemical Society

Nickel transition metal Chemistry nickel(II) Ni2+ complex ions ligand substitution redox chemical reactions principal oxidation states +2 +3 GCE AS A2 IB A level inorganic chemistry revision notes
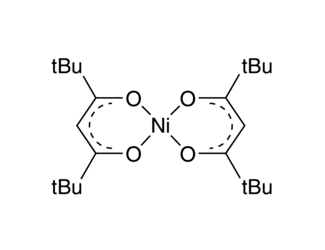
Organometallic Ni( ii ), Ni( iii ), and Ni( iv ) complexes relevant to carbon–carbon and carbon–oxygen bond formation reactions - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QI01486B

The idealized crystal structure of α-Ni(OH) 2 · xH 2 O represented by... | Download Scientific Diagram
![Explain hybridisation, geometry and magnetic property of [Ni(CN)4]^2 - ion using Valence Bond Theory(VBT). [Atomic number of Ni is 28 ]. Explain hybridisation, geometry and magnetic property of [Ni(CN)4]^2 - ion using Valence Bond Theory(VBT). [Atomic number of Ni is 28 ].](https://haygot.s3.amazonaws.com/questions/874625_947137_ans_e945a0328151431c8601af56eaa7dcb3.png)
Explain hybridisation, geometry and magnetic property of [Ni(CN)4]^2 - ion using Valence Bond Theory(VBT). [Atomic number of Ni is 28 ].

![Draw the structure of [Ni(NCS)2(PPh3)2]. Show the NCS bond. | Homework.Study.com Draw the structure of [Ni(NCS)2(PPh3)2]. Show the NCS bond. | Homework.Study.com](https://homework.study.com/cimages/multimages/16/marvinjs-output_281316354014965502313.png)

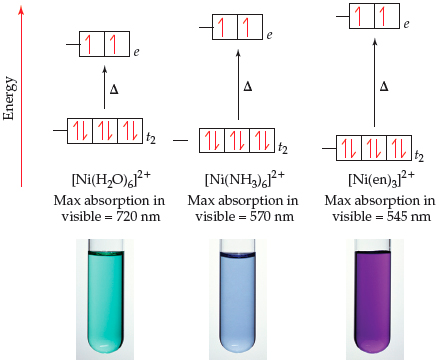


![Ni(en)3]2+ Ni(en)3]2+](http://www.chemtube3d.com/images/gallery/inorganicsjpgs/Nien32_.jpg)